Clinical Trials
MDS Clinical Trial Platform
MDSF is proud to partner with SparkCures, LLC, a patient navigation and technology company. We have selected to partner with SparkCures to develop this custom platform for MDS patients, caregivers, and healthcare providers because of their extensive experience in patient support and technology. This platform will offer fast and personalized clinical trial screening and matching services, simplifying the complex landscape of clinical trial options for MDS patients. This collaboration aims to revolutionize clinical trial navigation and empower MDS patients and their care partners to explore personalized clinical trial options.
What is a clinical trial?
Clinical trials study new interventions (drugs or procedures) to evaluate their safety and effectiveness in humans. Trials follow a careful set of steps, allowing for the systematic gathering of information to answer questions and confirm hypotheses that were formed earlier, in either laboratory experiments or preliminary trials.
Trials are in four phases:
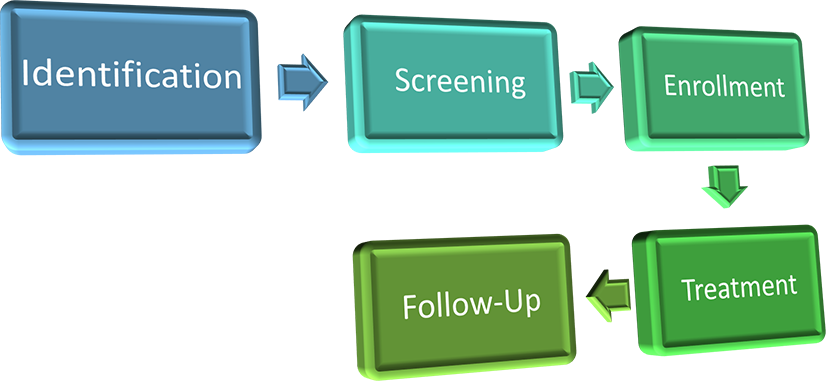
Screening
After a patient is identified and confirms they are ready to move forward, the patient will begin the screening process. The purpose of the screening process is to make sure that it is safe and medically appropriate for the patient to receive treatment on trial. Screening should always start with the informed consent process. Consents for clinical trials are often lengthy; it is important to read the whole document. Before signing the consent, the patient must be given the opportunity to read the document, have all questions answered by study staff, and have time to discuss the trial fully with family and healthcare providers if desired.
The consent will give information on the following:
After signing consent, the next step is to complete screening procedures. Each trial will require different tests at screening. These tests help determine if the patient is a good fit for the trial and the trial is a good fit for the patient; both have to be true to move forward.
Common screening procedures for MDS trials include the following:
After testing is complete, study personnel will complete an eligibility packet that goes over the criteria patients must meet to go on trial. After completion of this packet by the clinical trial site, it is sent to the sponsor or drug company for approval and final sign-off.
Enrollment
Once a patient is deemed eligible, they will be scheduled to begin treatment as part of the clinical trial. Some investigational medications are given in the hospital while others are given in the clinic. The decision on where to give the medication is made based on the safety profile of the medication, the need for monitoring, and the amount of study procedures required. Some medications are given in pill or tablet form and can be taken at home on certain days. The study personnel will go over all the details about the study medication to include how it is given, the administration schedule, and how to store the medication prior to the patient receiving the first dose. Patients should also receive contact information for issues that occur after hours or on the weekend. All paperwork to include pill diaries, required questionaries, and financial re-imbursement forms will be reviewed with the patient. Insurance approval is finalized prior to starting therapy as well.
Treatment
Every trial has a different schedule of assessments. Often the first couple months on trial are the busiest. Patients can expect to be seen by their healthcare provider more frequently and more study samples will need to be collected. As the investigational drug will not yet be FDA approved for the condition being studied, it is necessary to draw drug levels at set timepoints to determine exactly how much of the drug is absorbed and how long it remains in the patient’s system. Pharmacokinetic and pharmacodynamic testing is done to determine how the body impacts the drug and how the drug impacts the body. Many research procedures such as vital signs, lab collections, and electrocardiograms have to be done on a very tight time schedule. Consistency is the key to getting accurate data and, ultimately, to getting drugs FDA approved.
While on study, patients are monitored for potential side effects and response to therapy. It is very important for patients to be honest with the provider about how they are feeling and how they believe they are tolerating the investigational drug.
Treatment will continue until one of the following occurs:
Follow-Up
When the decision is made for a patient to come off trial, the patient will complete an end-of-treatment visit. The purpose of this visit is to identify any side effects that are ongoing and develop an appropriate follow-up schedule. Many trials require the patient to come in approximately 30-90 days after the last dose of study drug for a safety evaluation. If any side effects are lingering, there are often additional follow-up visits until these have resolved. Clinical trial follow-up also includes a long-term follow up period during which patients are contacted to review current therapy they are receiving and disease status. Just as with participating in the clinical trial, patients can opt out of the follow-up portion as well; however, that long-term data really helps with developing a side effect profile for the trial medication. It usually takes years for a clinical trial to wrap up. All published data from the trial is made available on www.clinicaltrials.gov.
Adoptive Cellular Therapy: Involves using the patient’s own immune system to fight cancer.
Allogeneic: Involving matched bone marrow or peripheral blood stem cells from a donor that are collected, stored, and then infused into a patient (recipient) following high-dose chemotherapy with or without radiation therapy. The cells cannot come from the patient needing the transplant.
Antibody drug conjugate: Therapy created by combining a monoclonal antibody with a cytotoxic agent.
BCL-2 Inhibitor: Programmed cell death is controlled by BCL-2 proteins. Issues within this family of proteins can lead to avoidance of pre-programmed cell death (apoptosis) and allow for the proliferation and extended survival of cancer cells. Inhibition of BCL-2 can lead to cancer cell death.
Blast Cells: Immature blood cells that would normally become fully functional mature red cells, white cells, or platelets. The number of blast cells in the bone marrow helps define how severe MDS is in a person. When 20 or more out of 100 cells in the bone marrow are blasts, this is considered acute myeloid leukemia (AML).
Bone Marrow Aspiration: The process of removing bone marrow from a specific area using a small needle and syringe. Used for diagnostic purposes. Tests may also be run on the bone marrow cells to look for any genetic abnormalities.
Bone Marrow Biopsy: The bone marrow biopsy is a small core (the shape and size of a medium pencil lead) of the spongy center of the bone marrow. It provides information about the cellularity of the bone marrow (crowded=hypercellular, empty= hypocellular). It will also provide useful information about iron storage, scarring (fibrosis), and the presence of any other abnormal cells.
CAR-T: Chimeric antigen receptor (CAR) T cell therapy involves removing the patient’s own immune cells, modifying them in the lab so that they recognize the patient’s cancer as foreign, and then infusing the cells back into the patient.
Chemotherapy: The use of medicines that kill cells (cytotoxic agents). People with high-risk or intermediate-2 risk myelodysplastic syndrome (MDS) may be given chemotherapy. Chemotherapy may also hurt healthy cells causing side-effects. If chemotherapy works in controlling abnormal cells, then relatively normal blood cells will start to grow again. Chemotherapy agents include: cytarabine (Ara-C) and hydroxyurea (Hydrea), daunorubicin (Cerubidine), idarubicin (Idamycin), and mitoxanrone (Novantrone).
Combination Chemotherapy: The use of more than one drug during cancer treatments.
Cytogenetics: Testing that is performed on bone marrow samples and examines the chromosomes of the cells. Your cytogenetic results are used to identify the type of MDS you have and to calculate the International Prognostic Scoring System (IPSS) and the revised IPSS (IPSS-R) risk category.
1. Deletion 5q- – deletion of chromosome 5
2. Deletion 20 – deletion of chromosome 20
3. Deletion Y – deletion of the Y chromosome
4. Monosomy 7 – loss of one of the two 7 chromosomes
5. Trisomy 8 – addition of a third chromosome 8
Erythropoietin-stimulating Agent (ESA): A medicine used to help the bone marrow make more red blood cells. Epoetin alfa (Epogen, Procrit) and darbepoetin alfa (Aranesp) are erythropoiesis-stimulating agents that can help boost the red blood cell count of some bone marrow failure patients. Also called red blood cell growth factor
Flow Cytometry: A laboratory test that gives information about cells, such as size, shape, and percentage of live cells. Flow cytometry is the test doctors use to assess for specific proteins on the surface of blood cells. It is the standard test for confirming a diagnosis of paroxysmal nocturnal hemoglobinuria (PNH).
Fluorescence In Situ Hybridization (FISH): An important laboratory test used to help doctors look for chromosomal abnormalities and other genetic mutations. Fluorescence in situ hybridization, also called FISH, directs colored light under a microscope at parts of chromosomes or genes. Missing or rearranged chromosomes are identified using FISH.
Hypomethylating Agent: A hypomethylating agent is a drug that inhibits DNA methylation. Works by preventing certain genes involved in controlling cancer from being silenced, allowing for the normal functioning of the tumor suppressor genes.
IPSS/IPSS-R: An International Prognostic Scoring System – system for grading the severity of MDS. The system turns patient data into a score. The score tells how quickly a myelodysplastic syndrome (MDS) case is progressing and helps predict what may happen with the patient’s MDS in the future.
Kinase inhibitor: Kinase inhibitors block the action of protein kinases. Through a process of adding a phosphate group to a protein (phosphorylation), protein kinases can turn on or off proteins which are needed for unregulated growth of cancer cells.
Myelodysplastic Syndromes (MDS): The Myelodysplastic Syndromes (MDS) are a group of bone marrow failure disorders. Myelo refers to the bone marrow. Dysplastic means abnormal growth or development. In MDS, the bone marrow does not make blood cells normally. The result is too few cells or low blood counts (cytopenias) and cells that do not function properly.
Monoclonal antibodies: Monoclonal antibodies are antibodies that target specific antigens such as those found on cancer cells.
Natural killer cells: A type of cell that lacks B-cell and T-cell receptors and attacks mutant and virus-infected cells.
Peripheral Blood Stem Cell (PBSC) Transplant: A procedure where stem cells are collected from the donor’s circulating (peripheral) blood. These stem cells are then given to the patient through an intravenous (IV) line. In time, donated stem cells start making new, healthy blood cells. Also called PBSC transplant.
Placebo: A placebo is an inactive pill, liquid, or powder that has no treatment value. Placebo use in clinical trials is extremely uncommon today.
Refractory: Not responsive to treatment or cure. For example, refractory anemia is a low red blood cell count that doesn’t respond to standard treatments.
Ring Sideroblast: A red blood cell that has too much iron. The iron typically forms a ring around the cell’s nucleus.
Telomerase inhibitor: Telomerase is an enzyme that is responsible for adding telomeres to DNA strands. Cancer cells are known to have overactive telomerase which protects the cancer cells and allows them to proliferate. Telomerase inhibitors function by inhibiting the enzyme telomerase.
How does insurance work while I’m on a clinical trial?
It is best to think of the insurance company and the sponsoring drug company as partners. The cost of procedures, tests, and medications you would receive for your MDS routinely as part of standard of care, meaning even if you were not participating in a clinical trial, will still be billed to your insurance like normal and co-pays will still apply. The costs of procedures, tests, and medications you will only receive because of the clinical trial will be billed to the sponsoring drug company. It is the responsibility of the research staff to check-in with your insurance to ensure they will continue to pay for standard of care procedures while you are participating in a clinical trial. If you have questions about exactly how specific items are billed, reach out to the study team as they should be able to help.
Can I get paid to be on a clinical trial?
MDS treatment trials do not typically pay a patient to participate. There are sponsors who will pay or re-imbursement for travel related expenses to and from the clinical trial site. To be eligible for these funds, patients need to meet specific criteria such as living a certain distance from the medical center.
What are some of the patient’s responsibilities while on trial?
The number one thing to remember is to always be honest. Some patients are afraid that if they are experiencing side effects and they tell the clinic staff, they will be removed from the trial. While coming off trial is a possibility, it’s also possible that the dose of the drug may be decreased, or the investigational drug can be held for a time rather than discontinued. Your honesty ensures safety is the top priority.
Some of the other responsibilities include:
What does it mean when the doctor says a slot needs to be approved for me to go on trial?
For some trials, the clinical trial site must request a slot in order for a patient to participate. It gets confusing because the trial may be open, but not currently enrolling if all slots are taken. Slots are often used for phase I trials as different doses are studied in small groups or cohorts. Studies later in development will use slots as well if they are getting close to meeting the enrollment goal.
I don’t want to be a guinea pig. Is that what it is like being on a clinical trial today?
No. While it’s true that there are a lot of unknowns with clinical trials, participants are always kept in the loop when new information becomes available. If any new side effects or safety information is brought to light during the course of the trial, each participant is made aware in a timely manner and asked to sign an updated consent form with the new information included. Patients are carefully selected for trials. There’s no way to know ahead of time if a patient will respond to the therapy; however, patients are removed from trial if/when it becomes clear that they are not receiving benefit from the investigational product. Medical ethics are upheld and if there is ever concern that trial is no longer a good option for a participant, the provider will have that conversation with the patient to determine next steps.
It is important to note that there are now trials for first-line therapy. It is no longer a last resort. The MDS clinical trial landscape is a mixture of trials for people all along their MDS journey.
Can I still take my other medication while on a clinical trial?
During the screening process, your medication list will be reviewed to ensure that nothing you are currently taking will interact with the investigational drug. Your provider will explain any medication changes that would be needed for you to go on trial. If you need to start any new medications after going on trial, reach out to the study staff so they can review for potential interactions. Make sure to tell your provider about any herbal substances and vitamins you take as they can interact with study medications as well.
Do all clinical trials involve medications?
No. Treatment trials involve an intervention such as a medication or other type of therapy. Observational trials involve observation only and do not include any intervention. Observation trials may include quality of life questionnaires, sample collection, medical record review, and data collection.
Can I still see my other doctors while I’m on a trial?
For clinical trial required physical assessments, you will need to see a provider who is trained on the study. For other exams, you can continue to see your regular physicians. It is recommended that you tell your primary care physician and all specialists that you are participating in a clinical trial.
I just had a bone marrow biopsy. Why do I have to get another done for screening?
It is common for the sponsor of a trial to require samples to be collected and analyzed in their central laboratory. By using the same lab for all participants, it creates consistency in the results. In addition, some of the testing performed by the central lab is outside of what is considered routine care and cannot be completed at the clinical trial site. Some trials will not require a repeat marrow at screening if your previous one was done within a certain window of time, but that is dependent on the trial.